
Introduction
In medical device manufacturing, every component produced can directly affect a patient's health outcome. A misaligned surgical instrument, an implant with surface irregularities, or a diagnostic housing that fails dimensional specifications isn't just a quality issue—it's a patient safety risk. This reality makes precision, repeatability, and production speed requirements the industry cannot compromise on.
That's where CNC (Computer Numerical Control) machining fits in. It's become the manufacturing backbone of the medical device industry because it addresses all three demands at once.
Unlike manual machining, which introduces operator variability, CNC delivers sub-millimeter tolerances consistently across thousands of parts. It compresses development timelines through rapid prototyping in production-grade materials. And it provides the documented, traceable processes required to satisfy FDA regulatory standards — giving medical device manufacturers a reliable path through tight deadlines and stringent quality requirements.
TLDR
- CNC machining achieves tolerances as tight as ±0.0001 inches, essential for implants and surgical instruments where dimensional accuracy directly impacts clinical outcomes
- Supports both rapid prototyping and scalable production without re-tooling, compressing development cycles from 4-12 weeks to 1-4 weeks
- Titanium, stainless steel, and PEEK are all CNC-machinable to medical-grade standards using appropriate tooling and process controls
- Your machining partner's quality systems — inspection protocols, traceability, and process documentation — directly determine whether parts meet regulatory and clinical requirements
Why CNC Machining Is the Gold Standard for Medical Device Manufacturing
Manual machining cannot reliably achieve the dimensional consistency required for medical components. Human operators, no matter how skilled, introduce variability in toolpath execution, feed rates, and depth of cut. CNC machining eliminates this variability through computer-guided toolpaths that execute identically across every part in a production run.
Precision That Meets Medical Standards
Medical device tolerances typically range from ±0.0001" to ±0.001", far tighter than general industrial machining. High-precision CNC routinely achieves ±0.001" or better, while standard CNC holds around ±0.005".
That precision matters at the component level. Bone screw thread geometry must mate exactly with bone tissue, and implant bearing surfaces require micron-level smoothness to prevent wear debris generation — tolerances where manual methods simply cannot compete.
Traceability and Regulatory Compliance
CNC machining creates a fully documented, programmable process. Every toolpath, feed rate, spindle speed, and cut depth can be logged and archived. This documentation directly supports FDA 21 CFR Part 820 design control requirements and device history records. Under the new Quality Management System Regulation (QMSR), effective February 2, 2026, the FDA harmonizes 21 CFR Part 820 with ISO 13485, shifting documentation from traditional Device History Records (DHR) to the Medical Device File (MDF). CNC's inherent process documentation aligns perfectly with this regulatory evolution.
Repeatability at Scale
Once a CNC program is validated, it can produce thousands of identical parts without recalibration. A comparative study by MIT calculated rejection rates for a 50,000-unit production run: manual machining resulted in 6,155 scrapped units, while CNC machining resulted in 0. In high-volume medical component runs, every defect carries both financial and regulatory consequences. That consistency is what makes a validated CNC program one of the most valuable assets in a medical manufacturer's process library.
Why CNC Outperforms Alternatives
For structural and functional medical parts, CNC machining outperforms both manual methods and additive manufacturing where material density, surface finish, and mechanical performance are non-negotiable. The comparison with 3D printing is particularly relevant as adoption grows.
| Property | CNC Machining | Additive Manufacturing (Ti-6Al-4V) |
|---|---|---|
| Surface roughness (Ra) | Below 0.1µm (as low as 0.010µm) | ~3µm typical minimum |
| Fatigue resistance | Full wrought-material properties | Often reduced due to microstructural differences |
| Material density | Fully dense | Variable; porosity risk |
| Best use case | Articulating surfaces, load-bearing implants | Complex internal geometries |

3D printing has legitimate applications in medical device development, particularly for patient-specific or geometrically complex parts. For articulating joint surfaces requiring Ra 0.010–0.015µm to prevent wear debris, CNC remains the only reliable path.
Key Applications: What Medical Components CNC Machining Produces
Surgical Instruments
CNC machining produces scalpels, forceps, retractors, and bone saws with the edge geometry, surface finish, and dimensional accuracy required for both single-use and reusable surgical tools. Key requirements include:
- Surface finish: Ra < 0.8 µm to prevent bacterial biofilm formation
- Cutting edge geometry: Consistent angles held to tight tolerances across production runs
- Grip and articulation: Textured surfaces and joint mechanisms that perform reliably under clinical stress
Orthopedic and Implantable Devices
CNC machines hip and knee implants, spinal implants, and bone screws to tolerances that manual fabrication cannot reliably hit. An estimated 95% of all bone screws are made on Swiss turning machines, which use a guide bushing to support material close to the cutting tool — minimizing deflection and enabling micron-level accuracy for long, slender parts with high length-to-diameter ratios. Swiss lathes hold typical tolerances of ±0.0001" to ±0.0005", essential for bone screw threads that must mate precisely with bone tissue.
Bearing surfaces are equally demanding. Articulating joint surfaces require Ra 0.010–0.015 µm to prevent wear debris that can trigger inflammatory responses — a finish specification that defines the boundary between a functioning implant and a failed one.
Diagnostic and Imaging Equipment Housings
Beyond implantables, CNC machining also produces the structural enclosures, frames, and brackets that house MRI components, imaging systems, and patient monitoring equipment. These parts require dimensional accuracy for precise alignment of optical or electromagnetic systems and often need EMI shielding properties. Aluminum and stainless steel housings are machined to tight flatness and perpendicularity tolerances, ensuring diagnostic equipment maintains calibration over years of clinical use.
Custom Prosthetics and Patient-Specific Devices
For patient-specific devices, CAD-driven CNC machining takes customization further than standard production allows. A surgeon orders a prosthetic based on the patient's CT or MRI scan, and CNC programming converts that scan data into toolpaths within hours. What once took weeks manually now ships in a single day — compressing time-to-surgery and improving outcomes through better anatomical fit.
Micro-Manufactured Components
High-RPM micro-machining produces the tiny gears, connectors, and internal mechanisms found in pacemakers, drug delivery systems, and minimally invasive surgical tools. Micro spindles achieve speeds up to 120,000 RPM, delivering medical-grade accuracy for features measuring 100–500 micrometers.
The dimensional targets at this scale leave almost no margin for error:
- Ceramic pacemaker feedthroughs: via holes of ~25–100 µm, pin diameters of 25–50 µm
- Micro surgical forceps tips: serrations 150 µm wide with 50 µm depth
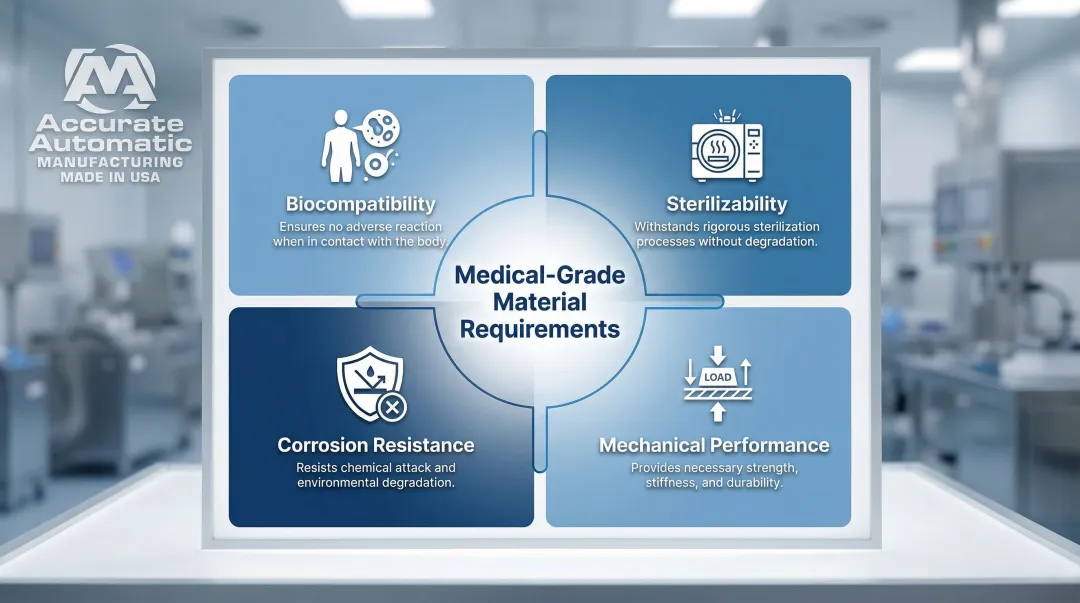
Materials That Meet Medical-Grade Requirements
Medical CNC machining materials must meet four primary criteria: biocompatibility (safe for human tissue contact), sterilizability (autoclave resistance), corrosion resistance (withstands bodily fluids), and mechanical performance under in-body or clinical conditions.
Titanium Alloys
- Primary alloys: Ti-6Al-4V ELI (ASTM F136) and unalloyed titanium (ASTM F67)
- Properties: Lightweight, strong, biocompatible, and elastically similar to bone
- Applications: Ideal for implants and orthopedic devices
- Elastic modulus: 105–116 GPa (~5× stiffer than cortical bone) — reduces stress shielding risk compared to stainless steel
- Machining notes: Ti-6Al-4V ELI's low thermal conductivity concentrates heat at the cutting tool. Recommended milling speeds are 50–70 m/min; uncoated carbide tools handle high temperatures well. Minimum Quantity Lubrication (MQL) reduces friction and improves surface quality over flood cooling.
Stainless Steel (300-Series)
- Primary grades: Type 316L (ASTM F138) designed for surgical implants
- Market share: The surgical instruments segment held approximately 60% of the global surgical stainless steel market in 2024
- Properties: Ease of machining, oxidation resistance, and autoclave compatibility
- Applications: Instruments, housings, and structural components; 316L's low carbon content (below 0.03%) supports biocompatibility for temporary orthopedic implants
- Elastic modulus: 190–200 GPa (~10× stiffer than cortical bone) — limits use in permanent load-bearing implants but suits rigid surgical instruments
- Machining notes: Post-machining passivation is required to restore corrosion resistance after cutting operations.
Medical-Grade Plastics (PEEK, Delrin/Acetal)
- PEEK (ASTM F2026): Elastic modulus of 3–4 GPa, closely matching bone — ideal for spinal fusion cages and load-sharing applications where stress shielding must be minimized
- Delrin/Acetal (ASTM F1855): Non-conductive, non-metallic option for components requiring weight reduction, electrical isolation, or radiolucency; restricted to non-implant applications until biocompatibility evaluations are completed
- Applications: Surgical instrument handles, diagnostic equipment housings, and temporary orthopedic components
Aluminum Alloys
- Applications: Cost-effective and lightweight for enclosures, carts, and structural frames that don't require biocompatibility
- Post-processing: Often anodized for corrosion resistance and EMI shielding
Each material choice cascades into specific machining decisions: cutting speeds, tooling coatings, coolant strategy, and post-processing steps. Getting those parameters right is what separates a dimensionally accurate medical component from a costly nonconformance.
How CNC Machining Accelerates Medical Device Production
CAD-to-Machine Workflows
Once a CAD model is validated, CNC programming can begin immediately, eliminating the 4-12 week tooling lead times associated with injection molding. CNC machining delivers parts in 1-4 weeks, while injection molding requires building molds first, adding 4-12 weeks minimum. For projects requiring fewer than 100 pieces, CNC machining is the better option. The cost break-even point between injection molding and CNC typically falls around 1,400 units, making CNC highly economical for early-stage production and clinical trials.
Rapid Prototyping Speed
CNC machining produces functional, production-representative prototypes from the final intended material. This means engineering validation, fit testing, and regulatory submissions can proceed faster.
A medical device engineer can test a titanium implant prototype machined from ASTM F136 material, confident that mechanical properties and surface finish match what will be used in production. This eliminates the risk of surrogate material testing that fails to accurately predict clinical performance.
Multi-Axis Machining as a Production Accelerator
Multi-axis machining (3-axis, 4-axis, 5-axis) allows complex geometries to be completed in fewer setups, reducing handling time, fixturing errors, and overall cycle time. Transitioning from 3-axis to 5-axis machining reduces required setups by 60% (from 5 setups to 2) and cuts cycle times by up to 30% (from 48 minutes to 33 minutes), while eliminating compounding fixturing errors. Complex orthopedic implants and surgical instruments with freeform surfaces demand exactly this kind of setup reduction to hold tight tolerances across the full geometry.

Automation's Role in Throughput
Automated pallet changers, lights-out machining, and reduced operator intervention allow medical CNC shops to run high-volume or complex jobs continuously without sacrificing quality or adding labor costs. Traditional manual machine tending yields only 40–55% spindle utilization, while robotic tending pushes utilization to 85–92%, adding 40–60 hours of productive time per week. In a medical device company machining orthopedic implants, robotic tending increased throughput by 47% (producing 280 implants per day vs. 190 manually).
Scalability Without Re-Tooling
CNC machining bridges the gap between low-volume prototype runs and high-volume production without re-tooling. This flexibility is particularly valuable during iterative device development or regulatory change cycles:
- Initial testing: 10 prototypes from the same program
- Clinical trials: scale to 100 units without new tooling
- Market launch: ramp to 10,000 units with only minor process refinements
A contract machining partner like Accurate Automatic MFG, with capabilities across CNC milling, turning, and screw machine operations, can support medical device manufacturers through each of these production stages without restarting from zero.
Quality Control and Regulatory Compliance in Medical CNC Machining
Multi-Stage Quality Control Process
Medical CNC machining uses in-process inspection — Coordinate Measuring Machines (CMMs), laser measurement, and surface profilometers — to verify dimensional accuracy at each production stage before parts move forward. ISO 10360-2 specifies acceptance tests for verifying CMM performance.
In automated medical cells, in-cell CMM gauging (such as the Renishaw Equator) checks critical dimensions on every part. When a dimension trends out of tolerance, the system automatically adjusts CNC tool offsets to compensate for tool wear — maintaining Cpk > 1.67 without operator intervention.
Documentation and Traceability Requirements
Medical device manufacturers must maintain material certifications, first article inspection (FAI) reports, and device history records that support FDA 21 CFR Part 820 quality system regulations. Confirm your machining partner maintains full material traceability from raw material certification through finished component delivery. Under the QMSR framework effective February 2, 2026, the Medical Device File (MDF) becomes the central production file, replacing legacy DHF/DMR/DHR terminology.
Post-Machining Processes for Medical Compliance
Documentation and traceability cover the paper trail — but physical compliance depends equally on what happens to parts after machining. Four post-machining steps determine whether a component meets clinical standards:
- Passivation removes iron particles and foreign materials embedded during machining. ASTM A967 covers general stainless steel passivation; ASTM F86 applies specifically to surgical implants requiring enhanced corrosion resistance.
- Surface finishing must meet tight roughness targets — Ra < 0.8 µm for surgical instruments to prevent bacterial biofilm formation, and Ra 0.010–0.015 µm for articulating joint surfaces to prevent wear debris.
- Deburring ensures precise edge geometry with no burrs that could cause tissue trauma during surgical use.
- Cleaning removes machining oils, particulates, and contaminants before components enter clinical environments.

What to Look for in a Medical CNC Machining Partner
Key Capability and Process Criteria
- Operates 4-axis or 5-axis machines capable of complex geometries, reducing setups and cycle times
- Has documented experience with medical-grade alloys (Ti-6Al-4V ELI, 316L stainless steel) and engineering plastics (PEEK, Delrin), including appropriate tooling and process controls
- Holds an established quality management system — ISO 9001 minimum, ISO 13485 preferred — with documented procedures covering material traceability, first article inspection (FAI), and process validation
- Offers Design for Manufacturability (DFM) feedback early in development, catching manufacturing challenges before toolpaths are programmed
Culture and Reliability
In medical manufacturing, a supplier's commitment to workmanship integrity, on-time delivery, and transparent communication matters as much as machine capability. Mistakes at the manufacturing stage can delay FDA submissions, trigger recalls, or affect patient outcomes. The right partner understands that stakes in this industry are not abstract — they're clinical.
Accurate Automatic MFG is a precision contract manufacturer offering CNC milling, CNC turning, and screw machine capabilities, with a focus on quality, workmanship, and relationships built on integrity. Their team is equipped to support medical device manufacturers who need a dependable machining partner. Contact them at sales@accurateautomaticmfg.com or +1 330-435-4575 to discuss your project requirements.
Frequently Asked Questions
What tolerances can CNC machining achieve for medical device components?
High-precision CNC machining routinely achieves tolerances of ±0.001" or tighter, with critical components like cardiovascular stents and orthopedic implant interfaces holding ±0.0001" (2.54 micrometers). This level of precision is necessary because dimensional deviations can affect implant fit, articulation mechanics, and long-term clinical performance.
What materials are most commonly used in CNC machining for medical devices?
The most common materials are titanium alloys (Ti-6Al-4V ELI), 316L stainless steel, PEEK, and aluminum alloys. Selection depends on the component's biocompatibility requirements, sterilization method, and mechanical load demands — each material serves a distinct clinical application.
Can CNC machining support both prototyping and full production of medical devices?
Yes. CNC machining scales directly from single prototypes to high-volume production using the same validated program — no re-tooling required. This continuity is valuable across iterative development cycles, from initial testing batches through clinical trials to full market launch.
How does CNC machining support FDA compliance in medical manufacturing?
CNC's documented process creates a traceable record of every toolpath, feed rate, and cut — directly supporting FDA 21 CFR Part 820 and the QMSR framework's Medical Device File (MDF) requirements (effective February 2026). This built-in traceability simplifies design control and device history record obligations throughout the product lifecycle.
What types of medical devices are most commonly manufactured using CNC machining?
The primary device categories include surgical instruments (scalpels, forceps, retractors), orthopedic implants (hip and knee implants, spinal implants, bone screws), diagnostic equipment housings (MRI components, imaging systems), custom prosthetics, and micro-manufactured components for pacemakers and minimally invasive surgical tools.
How does CNC machining compare to 3D printing for medical device manufacturing?
CNC machining outperforms 3D printing on material density, surface finish (Ra 0.1µm vs. Ra 3µm), and mechanical strength. For complex internal geometries or porous osseointegration structures, 3D printing has the advantage. Many implants use both: additive manufacturing for porous scaffolds, CNC machining for articulating surfaces.


